LYMEPOLICYWONK: LymeDisease.org Endorses New ILADS Lyme Disease Guidelines
The Centers for Diseases Control estimates that more than 300,000 people contract Lyme disease each year. People with Lyme frequently experience long delays in obtaining an initial diagnosis, have poor access to healthcare and suffer a severe burden of illness. Those who are not diagnosed early have a worse prognosis; half become chronically ill.
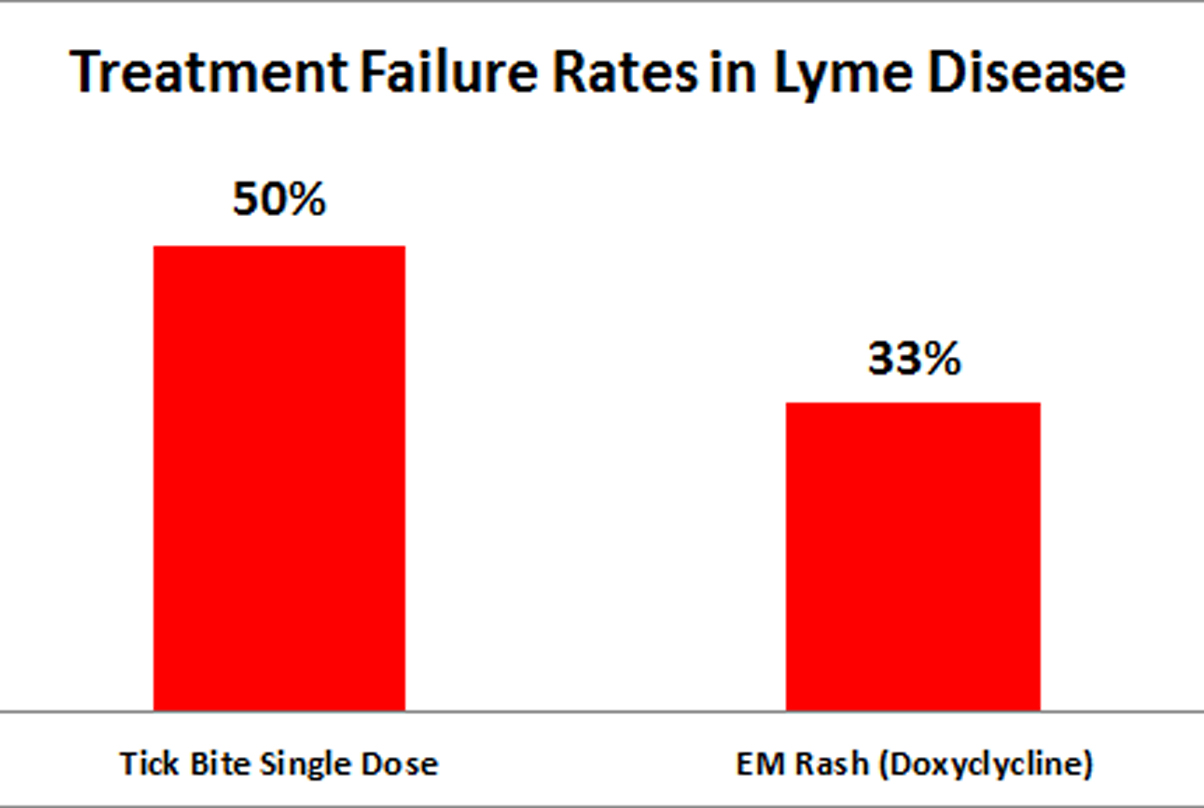
Focusing on how best to reduce the risk of developing chronic illness, the new guidelines target how to treat tick bites, erythema migrans rashes and persistent disease. Because the evidence base is poor, there is a strong need for clinical judgment and shared decision-making with patients. Many of the treatment decisions in Lyme disease depend on trade-offs. What are the risks and benefits for treating or not treating? How sick is the patient? How invasive is the treatment? To make informed medical choices patients need to understand the risks and benefits of treatment options. These guidelines provide that information.
The guidelines were developed over a three-year period and reflect the latest science. They are the first Lyme guidelines to use a rigorous systematic evidence evaluation approach called GRADE (Grading of Recommendations Assessment, Development, and Evaluation). GRADE has been adopted by other well-respected medical organizations including Cochrane Collaboration, the World Health Organization, and the National Institute of Health. The guidelines also reflect the high standards established by the Institute of Medicine for developing trustworthy guidelines and were reviewed by over 25 reviewers.
The complexity of Lyme disease makes a cookie cutter approach inappropriate noted Dr. Cameron, MD, MPH, lead author: “We moved away from designating a fixed duration for antibiotic therapy and instead encourage clinicians to tailor therapy based on the patient’s response to treatment.”
Coauthor Elizabeth Maloney, MD, commented on the need for clinicians to perform a deliberate and individualized assessment of the potential risks and benefits of various treatment options before making their initial selection: “We recommend careful follow-up because it allows them to adjust therapy as circumstances evolve. This more selective approach should reduce the risk of inadequate treatment giving rise to a chronic illness,” added Dr. Maloney.
The full guidelines, “Evidence Assessments and Guideline Recommendations in Lyme disease: The Clinical Management of Known Tick Bites, Erythema Migrans Rashes and Persistent Disease” were published online today in the August issue of Expert Review of Anti-infective therapy and are available on line for free access.
LYME POLICY WONK is written by Lorraine Johnson, JD, MBA, the Executive Director of LymeDisease.org. Contact her at lbjohnson@lymedisease.org. On Twitter, she’s @lymepolicywonk.





