Lyme Basics
Lyme Disease Tests
Testing for Lyme disease is complex and often controversial. The most commonly used tests—ELISA and Western Blot—were originally adapted for public health tracking in the 1980s and 1990s, not for diagnosing individual patients. Despite their limitations, they remain part of the CDC’s recommended Standard Two-Tier Testing (STTT) protocol.
Sensitivity vs. Specificity in Lyme Testing
Lyme disease tests are often inaccurate. Two key measures help explain this:
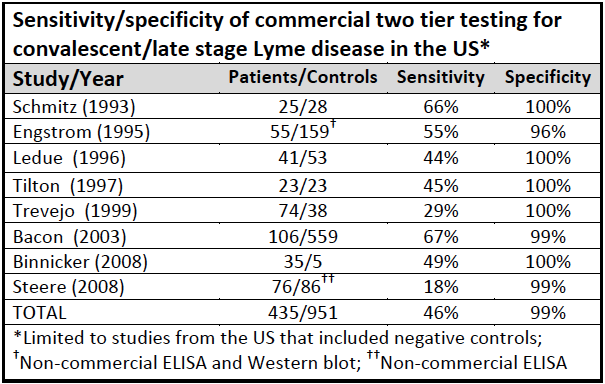
The ELISA test, typically used first, has particularly low sensitivity in early Lyme disease, when antibody levels are still building. Even the Western Blot, used to confirm ELISA results, misses many cases. One study showed it detected fewer than 54 out of every 100 confirmed infections.
Understanding Two-Tier Testing
The CDC recommends a two-step process:
Western Blot Criteria
Many symptomatic patients fall short of these thresholds, leaving them without a diagnosis or treatment.
Modified Two-Tier Testing (MTTT)
In 2019, the FDA cleared MTTT, a faster alternative that uses two ELISA-type tests instead of a Western Blot. It offers similar accuracy and easier interpretation.
As of 2024, the CDC recognizes both STTT and MTTT as valid. Labs like Labcorp have fully adopted MTTT, though STTT remains widely used. However, both methods can still miss true cases.
IGeneX offers FDA-cleared ImmunoBlot kits:
These tests detect more strains of Lyme bacteria than standard tests, improving sensitivity without sacrificing specificity. They’re the only kits that detect antibodies to OspA (31) and OspB (34). The CDC removed 31 and 34 from its Western blot guidelines in the early 2000s to avoid confusion with the Lyme vaccine, which was based on OspA (31). That vaccine was discontinued in 2002, but the exclusion remains — even though most people today were never vaccinated. As a result, some truly infected patients may test negative under CDC criteria simply because these bands aren’t counted.
Direct Detection Tests
Some tests aim to find the Lyme bacteria itself—not just your immune response:
Testing for Co-Infections
Ticks can carry multiple pathogens. Common co-infections include:
Because symptoms overlap, doctors may order additional blood tests. Early detection helps guide effective treatment.
The following labs offer Lyme tests that have been cleared by the FDA:
Testing for co-infections
Labs like IGeneX, Galaxy Diagnostics, and Vibrant Wellness offer comprehensive panels that screen for a range of tick-borne co-infections, including:
Babesia, Bartonella, Ehrlichia, Anaplasma, Rickettsia, Borrelia miyamotoi, and more.
These are laboratory-developed tests (LDTs)—created and used within a single certified lab. They’re regulated under CLIA, not the FDA, which means they haven’t gone through federal review. Even so, LDTs can help detect infections that standard tests might miss.

