IDSA: Caution when opening–contents under pressure

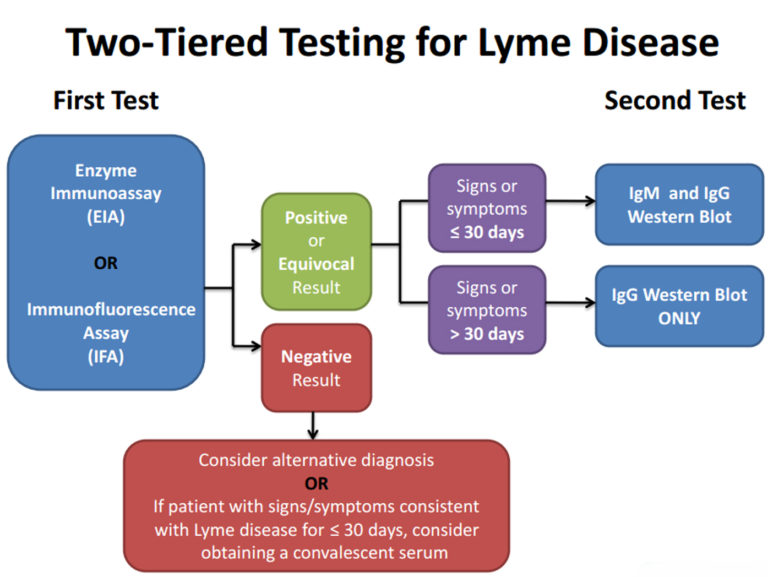
Dr. Paul Mead of the Centers for Diseases Control and Prevention (CDC) recently stated in the Wall Street Journal that the CDC’s recommended two-step process (which requires a positive ELISA test before a Western blot can be given) is accurate and was developed specifically to aid diagnosis of Lyme disease. A stunning assertion given that it directly conflicts with Dr. Mead’s previous testimony before the Connecticut Department of Public Health and the Attorney General’s office. It is also at odds with the CDC case definition for Lyme disease, which recognizes the validity of a stand-alone Western blot. This turnaround, coupled with LabCorp’s recent decision to abandon a stand-alone Western blot, has patients up in arms. And, rightly so.

During 2010, Dr. Stricker and I published nine articles or letters on Lyme disease. Shortly after the New Year, another publication by us came out. This post lists these 10 publications and tells you how to access those that are available without charge. More after the jump. . .
Yesterday, the CDC officially announced that it “recommends that laboratory tests cleared or approved by FDA be used to aid in the routine diagnosis of Lyme disease.” This is a shame. Waiting for FDA approval suppresses innovation in Lyme testing and furthers the interests of those who have vested interests in the current flawed lab tests which miss as many cases as they detect. Neither of these is good for patients.

Over 6,100 responded to our IDSA survey within one month! That’s YOU being engaged. Everyone who has worked on this project has been deeply touched by your responses. In addition to answering our survey questions, over 1,700 patients offered their thoughts in a tiny comment box. I have to say, I was unprepared for the depth of responses offered. I know that those of us who reviewed the comments teared up. You may too.

Biased reporting harms the credibility of journalism, harms patients, and misleads the public. The recent Tribune piece, which is now being republished in other Tribune outlets (including the Los Angeles Times) distorts and manipulates reality and makes a ‘good story’ at the expense of professional journalism. It does this by ignoring science, and by characterizing patients as hapless victims and their physicians as frauds. It was called to task for its profound lack of professionalism by Knight Science Journalism Tracker. Articles on science that do not present both sides of a legitimate controversy in science do a serious injustice and may violate the canons of journalist ethics established by the Society of Professional Journalism. Today, I look at some of those canons and point out how the Tribune article fell short. More after the jump. . .

CALDA, the national Lyme Disease Association and Time for Lyme have issued a joint press release regarding the IDSA Lyme review hearing, which is schedule for July 30th in Washington D.C. The release highlights the growing opposition to the IDSA guidelines, the importance of acknowledging the full spectrum of science in Lyme disease, and the importance of permitting clinical judgment and treatment options for patients. Concerns are also raised about the current process, which Attorney Lorraine Johnson points out is not impartial because IDSA controls the process and has selected both the panel members and those who may present before the panel. Of critical importance is that fact that no physicians who treat chronic Lyme disease were selected to sit on the panel. The testimony will be aired live over the internet on July 30th.